A little over 75 years ago, Sidney Farber started the first leukemia clinical trial that included children with acute lymphoblastic leukemia (ALL). This marked the dawn of meticulous scientific research for demystifying acute leukemia therapy and was methodically detailed in the book The Emperor of All Maladies by Siddhartha Mukherjee.1
Over the past 7 decades, scientific advancements have unraveled several types and subtypes of leukemia, provided a better understanding of leukemia biology and pathogenesis, created newer drug targets, and led to the approval of less toxic therapeutic agents. People with many types of leukemia go on to have a normal quality of life and survival duration comparable to that of the general population.2 These changes have been especially profound in the past 2 to 3 decades wherein leukemia genomics and genetic markers have been used to individualize tailored therapy.
Leukemia genomics is the study of the genetic background of leukemia that aims to elucidate the underlying variations and alterations in the chromosomes and genes that play a role in the genesis and progression of leukemia.
The outlook for people diagnosed with leukemia in 2024 is promising. The lifetime research of several physicians and scientists, the participation of individuals with cancer in clinical trials, and translational studies have completely changed the outcomes of several types of leukemia.
Improvements in diagnostic, therapeutic, and prognostic tools, along with enhanced supportive therapy for leukemia (eg, better control of infections, better blood product support, and a teamwork approach), have led to a paradigm shift in outcomes of patients with leukemia. In the subsequent sections we will discuss the different types of leukemia, focusing on the more common ones; highlight the changes in their therapy over the past few decades; and describe where we stand today.
The Acute Leukemias
Leukemias are traditionally divided into acute and chronic types based on the immediate severity of the illness and the acuity of the clinical presentation. The classical annotation of a “life-threatening” blood cancer is usually associated with the acute leukemias, which warrant immediate diagnosis and urgent initiation of therapy for all people with the disease.
The 2 most common types of acute leukemia are acute myeloid leukemia (AML) and ALL. The most commonly presenting symptoms in acute leukemias are increased weakness, shortness of breath, bleeding from the gums, bruises on the skin, and swelling of lymph nodes in the neck, axilla, and groin; however, asymptomatic people may be diagnosed based on abnormal findings in their routine blood work.
Clinical Trials
Clinical trials are strictly supervised and monitored scientific investigations to study the tolerability and/or efficacy of new therapeutic or prophylactic agents in humans, primarily with the aim to mitigate diseases. There are different phases of a clinical trial, usually beginning with studying whether the agent is safe and tolerable and going all the way to comparing the efficacy of the drug with that of other available agents.
Translational Studies
Translational studies use laboratory-based research to investigate newer targets for therapy in conditions for which drugs can be developed. They also explore reasons why a drug would work differently in patients with the same leukemia/ disease. Translational studies often form the framework for drug discovery that eventually leads to clinical trials.
Acute Myeloid Leukemia
AML is an acute cancer of the myeloid family of cells, a subgroup of white blood cells. This type of cancer is rare, with around 20,000 new cases in the United States annually.3 The median age of diagnosis for AML is roughly 70 years, although younger folks (including children) can also develop this disease.
AML is quite diverse, with several genetic subtypes under one umbrella. Modern research has unearthed different genomic subgroups of the leukemia, which define initial presentation, choice of therapy, prognosis, and the need for allogeneic stem cell transplantation (allo-SCT) for long-term disease control.
Traditionally, intensive chemotherapy has been used to treat AML in fit, younger (usually age <65 years) patients, while low-intensity therapy has been used for older, unfit patients, followed by allo-SCT when needed. Intensive chemotherapy regimens use a combination of drugs associated with slightly higher risks of infection, and so adequate organ function and physical fitness are needed to tolerate the therapy. Much of the incidence of complications and toxicity tied to intensive chemotherapy is dependent on the center where therapy is administered, and on the facility’s infection control practices, supportive care services, blood bank, etc.
Over the past 2 decades, around 10 new drugs have been approved to treat AML, the majority of which are promising targeted therapies given alone or in combination with other drugs. While some of these treatments can be used across the spectrum of AML, the majority are genomically inspired and require particular genetic aberrations to be present for their use (eg, FLT3 inhibitors require FLT3 gene aberrations; IDH1 or IDH2 inhibitors require IDH1 or IDH2 gene mutations).
A particularly important drug developed through translational research and subsequently used in clinical trials led by Courtney D. DiNardo, MD, and Marina Konopleva, MD, PhD, at MD Anderson Cancer Center (MDACC) is venetoclax, which has had a far-reaching effect on modern AML therapy.4 When added to low-intensity therapy (eg, a hypomethylating agent that blocks certain pathways that help cancer cells grow) or chemotherapy, venetoclax almost doubled response and survival rates in older, unfit people with newly diagnosed AML; these findings led to the approval of venetoclax for use in this setting.4 Venetoclax, a well-tolerated oral drug, is now being used in combination with intensive chemotherapy, where it is furthering the effect of these treatments in fit, younger folks with AML.
In addition, for patients who need but are unable to proceed to allo-SCT because of factors including advanced age, comorbidities, or lack of a donor, these targeted agents (and some low-intensity, nontargeted agents) serve as maintenance therapy after the leukemia is in remission to extend the duration of remission and potentially cure a proportion of individuals.
Steps toward improvement in AML outcomes have been small and steady. Each new drug approval adds to the treatment options, and these newer options are geared toward more tolerable therapy than traditional chemotherapy drugs. The use of these new agents has catapulted some traditionally high-risk subtypes of AML to a more favorable status (eg, the case of FLT3-mutated AML, which was previously considered adverse-risk but is now considered intermediate-risk due to the use of potent FLT3 inhibitors).5,6 Coupled with the availability of new drugs, allo-SCT practices have also evolved over time, and at MDACC all patients up to 70 to 75 years of age undergo assessment for allo-SCT feasibility if their subtype of AML warrants.
In addition, we can better monitor the state of response to therapy using novel tools that detect residual leukemia cells in the bone marrow, the site of origin of the AML, often up to a level of detection of 1 leukemia cell among 10,000 to 1 million normal cells. These methods of minimal/measurable residual disease (MRD) assessment add to the prognostic algorithm and allow us to better monitor and predict people with a higher risk for relapse.
Despite these promising advancements, AML remains a relapse-prone disease, and certain genomic subtypes of AML (eg, AML with TP53 gene mutation) have very short durations of response and survival. Constant innovations toward new therapy approaches, including immunotherapy (using drugs to activate the self-immune system against leukemia) and cellular therapy (genetically modifying the patients’ immune cells to better detect and kill leukemia cells), are being actively pursued and are poised to make major positive strides.
In a population-based Surveillance, Epidemiology, and End Results analysis in the United States, the 5-year survival rate for AML has steadily increased from 9% in 1980 to 1989 to 28% in 2010 to 2017 (in individuals aged 15-39 years, it has increased from 24% to 63% over the same time points), highlighting the steady and incremental improvement in outcomes.7 At MDACC, the majority of people with AML are treated in clinical trials with novel agents using an intensive or low-intensity backbone based on recipient age and fitness. Optimizing the best therapy in frontline settings is probably the single most important intervention to garner the deepest and longest remission; relapsed/refractory (R/R) AML remains a challenge, though major improvements in outcomes are being observed in such settings as well.
A very interesting subtype of AML is acute promyelocytic leukemia (APL), which is generally defined by an aberrant exchange between chromosomes 15 and 17 (that juxtaposes the PML gene to RARα gene) and has a particularly high risk of bleeding manifestations (and rarely blood clots) at presentation. Using mostly nonchemotherapy approaches, this disease is possibly the most curable subtype among any AML and poses few long-term complications, especially after the initial phase of therapy is correctly circumnavigated. Every patient with AML needs to be accurately screened by pathologists to rule out APL, given the completely different treatment approach for this subtype of AML and its >90% cure rate, which should not be compromised at any cost through misdiagnosis or mistreatment.
Acute Lymphoblastic Leukemia
The second type of acute leukemia is the ALLs. These types of leukemias are more common in children; and for most younger patients with ALL, treatment is curative and leads to survival comparable to the normal population. Notably, this was the first leukemia for which the concept of combination chemotherapy was evaluated in clinical trials from the middle of the past century. Overall, this leukemia is even rarer than AML, with around 7000 new cases estimated to be reported in the United States in 2024, 60% of which are expected in people aged <18 years.3
ALL is a cancer of the lymphoid family of white blood cells. The 2 broad subtypes of ALL are T-cell ALL and B-cell ALL based on the subtype of lymphoid cells that have become cancerous. The traditional treatment approach has been the use of combination chemotherapy to bring the disease into remission, consolidate that remission through further cycles of chemotherapy, and then continue maintenance with lower intensity chemotherapy agents for 2 to 3 years. This approach has led to astounding success in most pediatric patients with ALL, but in adults (particularly older adults) survival outcomes have trailed behind. Two major reasons for this discrepancy are high frequency of adverse-risk genetic changes in older versus younger individuals and the inability of older folks to tolerate intensive chemotherapy because of heightened toxicity. Thus, older adults had to receive attenuated doses of chemotherapy and omit certain agents, which ultimately led to inferior long-term outcomes.
Research, particularly in B-cell ALL, has exposed drug targets that have led to a major change in treatment practices. Use of less-intensive chemotherapy combined with these targeted agents could be used in younger or older adults with improvement in outcomes.8 Two such drugs approved through clinical trials led by MDACC’s Hagop Kantarjian, MD, are inotuzumab ozogamicin (an artificial antibody that targets the protein CD22 in B-cell ALL) and blinatumomab (an immunotherapeutic antibody that enables immune cells to target the CD19 protein in B-cell ALL). These agents, first studied in patients with B-cell ALL that was R/R, improved survival over therapy with traditional chemotherapeutic agents.9,10
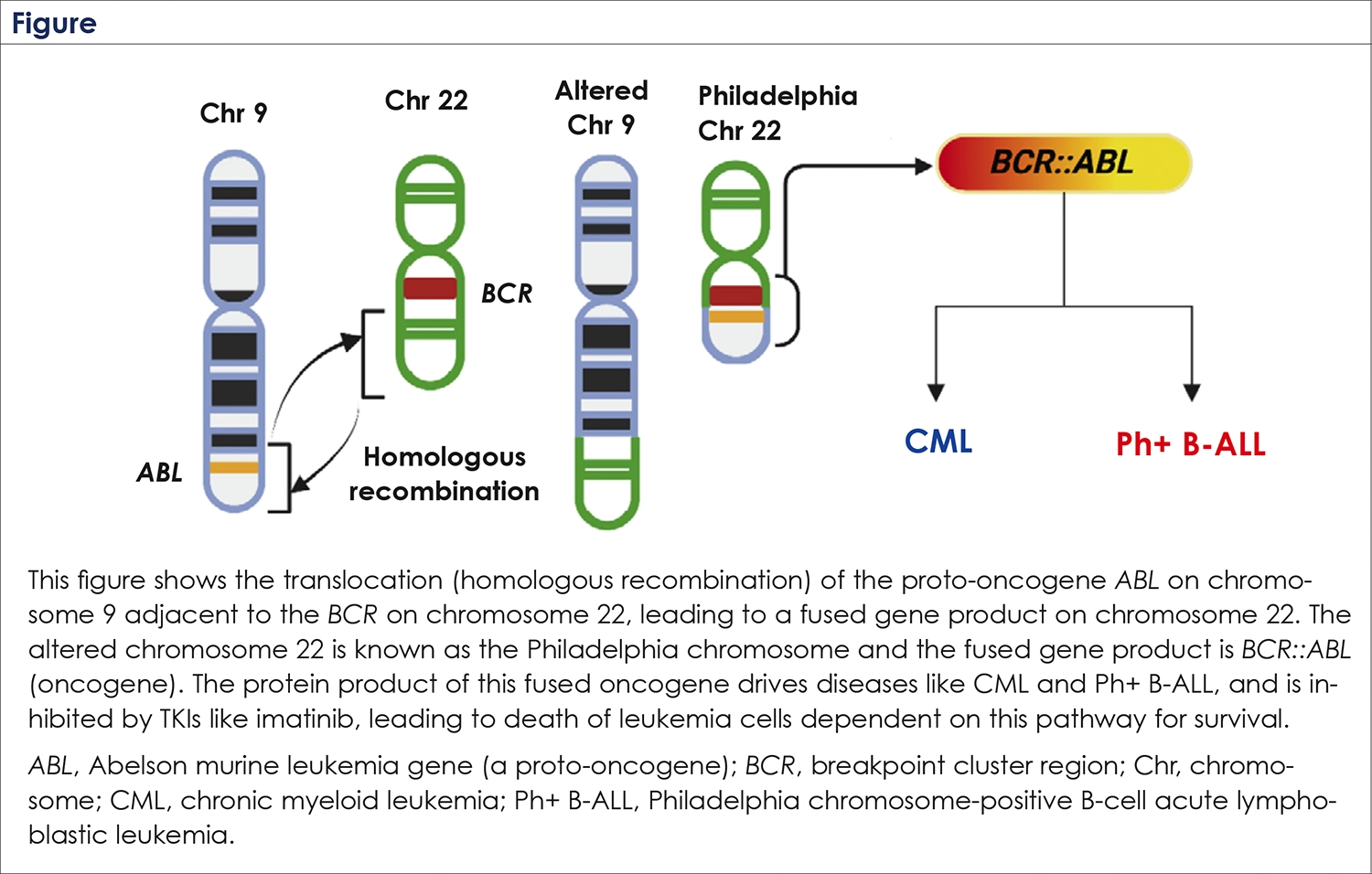
MDACC pioneered the use of a combination of these drugs with chemotherapy, significantly improving response rates and survival in the frontline and R/R settings, and these practices are now being followed across the world.11 A particular subtype of B-cell ALL, called Philadelphia chromosome-positive (Ph+) B-cell ALL has been at the forefront of these therapeutic innovations. Ph+ ALL is defined by the presence of a chromosomal aberration that juxtaposes a part of chromosome 9 onto chromosome 22, leading to activation of aberrant fusion of 2 genes (BCR and ABL) present on these chromosomes that drive this leukemia.
While traditionally considered a high-risk subtype of B-cell ALL that warranted allo-SCT once in remission, new frontiers of chemotherapy-free approaches (including the immunotherapy drug blinatumomab) have led to astounding survival outcomes, in studies led by MDACC’s Elias Jabbour, MD.12 These combinations of targeted therapies lead to quicker and deeper remissions and can be advocated to all adults, diluting the influence of intensive chemotherapies of earlier periods. A very interesting progress in therapy of B-cell ALL includes the use of cellular therapy approaches, also known as chimeric antigen receptor T-cell (CAR-T) therapy. In this innovative technique, the patient’s own T-cells are removed and then genetically engineered outside the body to allow for better recognition of proteins expressed in B-cell ALL and to enhance T-cell–mediated cell death.
These cells are then reintroduced back into the patient’s body after some chemotherapy; this approach, which is now approved by the FDA for R/R B-cell ALLs in children and adults, has opened newer avenues of therapy and is now being studied in clinical trials as frontline treatment after chemo-immunotherapy to reduce treatment duration.13
Along with these therapeutic innovations there has been tremendous progress in understanding the genomic risk factors in ALL and identifying people who have higher risks for relapse. As a testimony to these constant developments, the 5-year survival rate for adults with B-cell ALL treated at MDACC has increased from 24% in the 1980s to 66% in 2010-2022.8 In addition, the 5-year survival rate for Ph+ B-cell ALL has increased from 9% before 2000 to almost 90% in the past decade.8
A second subtype of ALL is the T-cell ALL, which have not stayed far from these advances. Although treatment for this subtype is still largely dependent on conventional chemotherapy and targeted drug development has lagged behind, a combined approach of better genomic risk stratification, superior combinations of chemotherapy, and better disease monitoring has improved outcomes for T-cell ALL. Data from MDACC show improvement in 5-year survival rates from 29% in the 1980s to around 60% in the past decade.8
We can now precisely monitor the extent of minimal/measurable residual disease assessment with therapy using sophisticated methods that can detect 1 leukemia cell among 1 million normal cells.
Across the spectrum of ALL, we can now precisely monitor the extent of MRD with therapy using sophisticated methods that can detect 1 leukemia cell among 1 million normal cells. This level of assessment helps us better understand early on how a patient is faring with therapy and facilitates monitoring patients post therapy to make further treatment decisions, including the need for allo-SCT.
The Chronic Leukemias
The chronic leukemias are defined by more indolent presentation and a less “life-threatening” initial clinical status. Patients with these disorders usually receive outpatient therapy, and most people with chronic leukemias have a normal quality and duration of life.
Chronic Myeloid Leukemia
Chronic myeloid leukemia (CML) is a slow-growing malignancy of the myeloid white blood cells that is usually diagnosed with regular blood work in asymptomatic individuals, although some folks have symptoms of weakness, bone pain, left-sided abdominal pain, or a dragging sensation from an enlarged spleen. The majority of people are not “sick” at diagnosis.
For those unfamiliar with the term “magic bullet,” it refers to targeted drug therapy for CML. In every sense, CML is the malignancy that has experienced the most radical change in outcomes in modern science. The story of tremendous success in drug development for CML started in 1959, when pathologist Peter C. Nowell and post-doctoral fellow David A. Hungerford of Philadelphia discovered a consistent chromosomal aberration in CML cells that resulted in a shortened chromosome 22 (humans have 23 pairs of chromosomes).14
Work by Janet Rowley and others in the early 1970s showed that the shortened chromosome 22 was secondary to the translocation of some of its material to chromosome 9 (Figure)15; a few years later she showed the chromosome 15 and 17 translocation to be pathogenomic for APL. Soon, researchers found that the gene transposed by the chromosomal change in CML was BCR (on chromosome 22) adjacent to ABL (on chromosome 9), creating a fusion gene (BCR::ABL).16 Subsequently, Owen Witte and colleagues presented seminal work on the aberrant continuous activation (oncogenicity) of the ABL gene (a proto-oncogene), instead of a normal on-off pattern, emanating from this fusion and its central role in CML.17 This unfolded very lucidly the biology behind CML and opened a field of work, along with similar specific aberrations discovered in other cancers, leading to the development of targeted drugs to block the underlying genetic aberrations in these cancers.
Despite the evolving knowledge of CML biology, until the end of the 1990s people diagnosed with this disease needed chemotherapy followed by an allo- SCT, and long-term survival was limited by the ability to receive allo-SCT. The therapies (chemotherapy or interferon) had significant side effects, which affected quality of life.
In the early 1990s, Nicholas Lydon, a biochemist working for what would later become Novartis, developed a small molecule called STI571 (which would later be called imatinib) that could inhibit the overactive ABL gene synthesized protein (ABL kinase); these drugs are anointed as BCR:ABL tyrosine kinase inhibitors (TKIs).18 The drug was soon tested, and after early phase trials established safety and selected the right dosing, imatinib was approved for use in CML in 2001.
In 2003 the phase 3 IRIS trial, which compared imatinib with low-dose cytarabine chemotherapy plus interferon alpha (the available standard-of-care treatment for CML), confirmed the superior efficacy of imatinib, and almost tripled the rates of complete cytogenetic response (clearance of the Ph+ chromosome after therapy).19 The astounding success of imatinib (a first-generation TKI) in CML led to the drug being referred to as the “magic bullet,” highlighting its potential to specifically affect leukemic cells with minimal damage to normal cells. Over the next decade, several second-generation TKIs (nilotinib, dasatinib, and bosutinib) and a third-generation TKI (ponatinib) have been studied in clinical trials, many of which were led by Dr. Hagop Kantarjian.20-22 The newer-generation drugs showed quicker efficacy and deeper responses than imatinib and even demonstrated activity in patients who developed imatinib resistance, although imatinib continued to hold its ground as a promising first-line therapy. There were astounding improvements in responses, and survival rates in CML in the post-TKI era are like those of the general population, with minimal to no adverse effect on quality of life.
Additionally, further research enables us to now categorize people with CML into different risk groups that help to ensure better monitoring practices and provide more precise prognoses.23 Moving on from the 1990s, when CML was one of the most common indications for allo-SCT, <5% of patients with CML today will need allo-SCT in their lifetime, the result of methodical research, drug development, and modern-day precision medicine. Some folks with CML develop advanced-phase CML, which behaves more like the acute leukemias. While this transformation to advanced-phase CML from the more docile, chronic- phase CML has been remarkably reduced following the introduction of imatinib and other TKIs, this is one niche area in CML where patients might need allo-SCT and intensification of therapy.24
TKIs like imatinib are pills that need to be taken daily and continuously. A question that seemed obvious was wondering if there could be a situation where these drugs could be stopped and the CML continue to remain in good control. While the initial therapeutic dictum was lifelong administration of these drugs, as long as tolerated, soon researchers started to understand trends where these TKIs could be stopped, and patients be stringently monitored thereafter. A concept now known as treatment-free remission (TFR) has grown increasingly popular in CML.25 A proportion of patients with CML who have had continued good depth of response with TKIs and are willing to be closely followed could be offered the option of TKI discontinuation after a few years of therapy. Though not every patient is eligible for TFR and some patients who are taken off the TKI could have disease resurgence and need to be restarted on the TKI, enabling more patients to reach this goal is a point of ongoing research in CML through use of more novel TKIs as well as studying combinations of TKIs with other agents.
The innovations in CML all the way from understanding the chromosomal and genetic changes, precise disease monitoring through assays that can check the molecular levels of the fused BCR::ABL culprit gene, to TKI development has been nothing short of a marvel in cancer medicine. More important have been the lives that have been saved and the ability of people to fully lead a normal life while on these oral pills, akin to therapy of more common ailments like hypertension or diabetes.
Chronic Lymphocytic Leukemia
Chronic lymphocytic leukemia (CLL) is an indolent malignancy of the B-lymphocytes and is the most common leukemia in adults. It is estimated that about 21,000 new cases of CLL will be diagnosed in the United States in 2024.26 The progress in CLL therapy has been revolutionary to say the least, with several new drug approvals over the past 10 years, all of them being targeted drugs. Additionally, the recent approval of a CAR-T therapy for R/R CLL has added another treatment option.
CLL is a slow-growing neoplasm. Around 20% to 30% of patients diagnosed with CLL will never need therapy for CLL in their lifetime, although it is difficult to accurately predict at diagnosis who these individuals are. People are usually diagnosed with CLL based on routine blood work showing elevated white blood cells (the lymphocyte component) or during evaluation of enlarged lymph nodes in the neck, armpits, and/or groin, although patients could have symptoms including fatigue, loss of appetite/weight, night sweats, and increased frequency of infections.
Because CLL is indolent, patients need to follow up with a hematologist/oncologist for symptom evolution, physical examination changes, and assessing parameters of the blood work which help decide when therapy needs to start. Individuals with CLL are at higher risk of infection given that the B-cells which are normally involved in antibody production and protection against certain infections are malignant and hypofunctional. Additionally, folks with CLL—even those who are just being observed and do not require therapy—need to be monitored for the development of other cancers (especially skin cancers, which are slightly more common in these individuals than in the general population).
A lot of research has gone into stratifying people with CLL into different risk groups. The primary role of this stratification is to guide the choice of therapy and long-term outcomes at the point of therapy initiation. Modern day CLL therapy has largely moved away from chemotherapy, relying heavily on targeted drugs and immunotherapy for almost all genomic subgroups of CLL. With this progress, the overall survival of patients with CLL now mirrors the survival of the general population. An important family of drug worthy of mention here are the Bruton tyrosine kinase (BTK) inhibitors. The BTK pathway is vital for B-cell development, and an overactive BTK pathway can lead to accumulation of malignant B-cells and to CLL. Ibrutinib was the first BTK inhibitor to be approved for R/R CLL in 2014.
In an important clinical trial led by Jan Burger, MD, PhD, at MDACC, ibrutinib showed superior potency over standard care in the first-line treatment of CLL; it was approved for CLL in frontline settings.
In an important clinical trial led by Jan Burger, MD, PhD, at MDACC, ibrutinib showed superior potency over standard care in the first-line treatment of CLL, leading to the approval of this drug for CLL in the frontline settings.27 As of 2024, 3 other BTK inhibitors with different toxicity profiles than ibrutinib have been approved for CLL. The newest of them, pirtobrutinib, can work in patients who become resistant to ibrutinib. BTK inhibitors continue to remain the cornerstone of targeted therapy in CLL, and other drugs like venetoclax have also been studied with success in CLL. The traditional dogma of BTK inhibitor therapy in CLL has been lifelong drug administration—so the obvious next question was to study time-limited therapy that could be administered for a fixed duration of time and then discontinued, with recipients remaining under observation and follow-up thereafter. To address this issue, different groups of drugs with potency in CLL were combined and studied, the foremost of which was the time-limited combination of a BTK inhibitor and venetoclax; the first such study was led by Nitin Jain, MD, and William Wierda, MD, PhD, at MDACC.28
Several other studies of this combination have cemented the efficacy and tolerability of this approach. Additionally, these studies showed the power of better CLL monitoring using MRD-based approaches, and a sizable fraction of people treated with such combinations continue to remain off therapy for significantly long durations without the disease becoming clinically re-apparent.
Monoclonal antibodies that target specific proteins on CLL cells have been around in CLL therapy for some time, but newer antibodies and their combination with BTK inhibitors and venetoclax are being studied in clinical trials as an “all-out” time-limited approach with the hope of better depth of disease clearance and longer time off therapy thereafter.
In the days of chemotherapy, R/R CLL was usually a challenging state and often needed allo-SCT for long-term disease control. With new drugs, the time to relapse has increased and even when relapse occurs there are several treatment options, including CAR T-cell therapy, that can be utilized.
Supportive therapy in CLL is extremely important, including screening for infections, timely vaccinations, immunoglobulin replacement in those with recurrent infections and low immunoglobin levels, screenings for other cancers, and addressing other concurrent comorbidities (eg, diabetes, hypertension). A combination of these modalities has made CLL a disease with very favorable outcomes, and people with the disease tend to have normal quality of life.
Challenges in Leukemia Therapy
Leukemia research has been a field of fascinating innovations at the bedrock of which lie individuals whose stories of success bear testament to the ever- improving treatment options and expertise, accessibility of cancer care, supportive therapy, and patient support groups, among others. This has been true for most countries across the world, but several challenges remain, some medical, some nonmedical.
Infections
The most common complication developing during cancer care, especially for acute leukemias, is infection. People receiving therapy for acute leukemias are extremely immunosuppressed both from the leukemia and the ongoing therapy, especially when their neutrophil counts are low. These risks significantly decline when the leukemia is in remission and neutrophil counts have recovered, but for people with R/R acute leukemias who undergo allo-SCT, the risks are even higher. The level of predisposition to particular infections varies depending on the disease type, therapy used, duration of neutropenia, etc. One of the most notorious groups of infections is fungal infections, which most commonly affect the lungs. The majority of patients on active acute leukemia therapy receive preventive antifungal medications, which have significantly reduced the risks of such infections.
Additionally, patients are also at risk for bacterial infections, or, rarely, reactivation of viral infections, although patients are usually covered with prophylactic therapies. Attention to infection prevention and rapid response to any new infection are indispensable in the care of people with leukemia. Notwithstanding the role of new drugs that fight leukemia better, advances in preventive and therapeutic antimicrobial medications and infection management have been a major cause for improvement in survival outcomes, especially among those with acute leukemias.
Transfusion Needs and Practices
Unlike industry-produced, scalable pharmaceuticals, blood products (eg, red blood cells, platelets) are completely dependent on voluntary human donation. Therapy for acute leukemias—and to a limited extent, chronic leukemias—is significantly dependent on transfusion support to maintain adequate levels of hemoglobin and platelets until the leukemia is in remission and the bone marrow recovers. Lessons learned during the COVID-19 pandemic showed the effect of constrained transfusion support through reduced voluntary donations and how it compromised adequate delivery of leukemia therapy. It is important for people to voluntarily donate blood to ensure adequate availability of blood products for patients with leukemia and other ailments whose therapy is largely contingent upon transfusion support.
Accessibility of Care
No cancer therapy, however efficacious, is of any benefit to the patient if it cannot be administered. Accessible cancer care beyond the echelons of top cancer care centers is important to permeate best practices to the community at large. Drug costs, especially of novel agents, have often been a deterrent for their use, and challenges with insurance approvals delay or prevent individuals from receiving the best therapy. Certain approaches, like Cost Plus Drugs, are actively trying to address the issues of costly cancer drugs, and this is welcome news.
Patients should also be made aware of the best therapy options available for their leukemia and referred when needed to academic centers where expertise on advanced leukemia therapy is available. Often, the best chance of controlling/curing leukemia is in the frontline settings, and the ability to advocate the best therapy at that time point (often in clinical trials) may significantly alter the course of therapy and long-term prognosis.
Goals of Care
Leukemia therapy has truly evolved in the past few decades, but despite novel agents and practices, therapy could still be associated with adverse events, hospitalizations, and loss of work. While this is more relevant for acute leukemias and R/R disease, physicians should always be cognizant about patient desires and the impact of their choice of therapy on quality of life and should discuss goals of care. The aim of leukemia therapy is curative or control with normalization of duration and quality of life, but sometimes these goals might be difficult to achieve. It is prudent that patients always have the scope to voice their informed choices and goals in discussion with their physicians and care team, so that a common path is chosen.
The Future Is Brighter
In just the past 12 months, around 6 drugs and one CAR T-cell therapy have been approved by the FDA as novel agents or drugs with expanded indications for different leukemias. This speaks volumes about the evolving landscape of leukemia therapy.
The aim of all these developments is to enable individuals not only to live longer but to live better and spend less time in the hospitals and more time doing things they love.
While this is reason to rejoice, certain high-risk leukemias continue to have inferior outcomes, and therapeutic research into them continues as we write this article. In the future, advancing research may help make therapies even more precise and of shorter duration, while enabling us to monitor patients better and curtail therapies in those with favorable risk disease or rapid/deep responses. The aim of all these developments is to enable individuals not only to live longer but to live better and spend less time in the hospitals and more time doing things they love.
As we reach the century mark beyond Sidney Farber’s first trial in leukemia, we hope also to be able to identify individuals at high risk for leukemias and prevent them from developing such cancers. And while deep dives into leukemia genetics are unfolding the unanswered questions of why one gets a leukemia, we can take pride in having achieved major strides thus far on how one develops the disease.
References
- Mukherjee S. The Emperor of All Maladies: A Biography of Cancer. Fourth Estate; 2011.
- Kantarjian HM, Jain N, Garcia-Manero G, et al. The cure of leukemia through the optimist’s prism. Cancer. 2022;128:240-259.
- National Cancer Institute Surveillance, Epidemiology, and End Results Program. SEER Cancer Stat Facts: Leukemia—acute lymphocytic leukemia. Accessed July 27, 2024. https://seer.cancer.gov/statfacts/html/alyl.html
- Döhner H, Wei AH, Appelbaum FR, et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022;140:1345-1377.
- Daver N, Schlenk RF, Russell NH, Levis MJ. Targeting FLT3 mutations in AML: review of current knowledge and evidence. Leukemia. 2019;33:299-312.
- DiNardo CD, Jonas BA, Pullarkat V, et al. Azacitidine and venetoclax in previously untreated acute myeloid leukemia. N Engl J Med. 2020;383:617-629.
- Sasaki K, Ravandi F, Kadia TM, et al. De novo acute myeloid leukemia: a population-based study of outcome in the United States based on the Surveillance, Epidemiology, and End Results (SEER) database, 1980 to 2017. Cancer. 2021;127:2049-2061.
- Jabbour E, Short NJ, Jain N, et al. The evolution of acute lymphoblastic leukemia research and therapy at MD Anderson over four decades. J Hematol Oncol. 2023;16:22.
- Kantarjian HM, DeAngelo DJ, Stelljes M, et al. Inotuzumab ozogamicin versus standard therapy for acute lymphoblastic leukemia. N Engl J Med. 2016;375:740-753.
- Kantarjian H, Stein A, Gökbuget N, et al. Blinatumomab versus chemotherapy for advanced acute lymphoblastic leukemia. N Engl J Med. 2017;376:836-847.
- Senapati J, Kantarjian H, Haddad FG, et al. SOHO state of the art updates and next questions | Next questions: acute lymphoblastic leukemia. Clin Lymphoma Myeloma Leuk. 2024;24:333-339.
- Jabbour E, Haddad FG, Short NJ, Kantarjian H. Treatment of adults with Philadelphia chromosome-positive acute lymphoblastic leukemia-from intensive chemotherapy combinations to chemotherapy- free regimens: a review. JAMA Oncol. 2022;8:1340-1348.
- Pasvolsky O, Kebriaei P, Shah BD, et al. Chimeric antigen receptor T-cell therapy for adult B-cell acute lymphoblastic leukemia: state-of-the-(C)ART and the road ahead. Blood Adv. 2023;7:3350-3360.
- Nowell PC, Hungerford DA. Chromosome studies in human leukemia. II. Chronic granulocytic leukemia. J Natl Cancer Inst. 1961;27:1013-1035.
- Rowley JD. A new consistent chromosomal abnormality in chronic myelogenous leukaemia identified by quinacrine fluorescence and Giemsa staining [Letter]. Nature. 1973;243:290-293.
- Heisterkamp N, Groffen J, Stephenson JR, et al. Chromosomal localization of human cellular homologues of two viral oncogenes. Nature. 1982;299:747-749.
- Lugo TG, Pendergast A-M, Muller AJ, Witte ON. Tyrosine kinase activity and transformation potency of bcr-abl oncogene products. Science. 1990;247:1079-1082.
- Druker BJ, Tamura S, Buchdunger E, et al. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr–Abl positive cells. Nat Med. 1996;2:561-566.
- O’Brien SG, Guilhot F, Larson RA, et al. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic- phase chronic myeloid leukemia. N Engl J Med. 2003;348:994-1004.
- Kantarjian H, Giles F, Wunderle L, et al. Nilotinib in imatinib-resistant CML and Philadelphia chromosome–positive ALL. N Engl J Med. 2006;354:2542-2551.
- Kantarjian H, Shah NP, Hochhaus A, et al. Dasatinib versus imatinib in newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2010;362:2260-2270.
- Cortes JE, Kim D-W, Pinilla-Ibarz J, et al. A phase 2 trial of ponatinib in Philadelphia chromosome–positive leukemias. N Engl J Med. 2013;369:1783-1796.
- Senapati J, Sasaki K, Issa GC, et al. Management of chronic myeloid leukemia in 2023 - common ground and common sense. Blood Cancer J. 2023;13:58.
- Senapati J, Jabbour E, Kantarjian H, Short NJ. Pathogenesis and management of accelerated and blast phases of chronic myeloid leukemia. Leukemia. 2023;37:5-17.
- Ross DM, Hughes TP. Treatment-free remission in patients with chronic myeloid leukaemia. Nat Rev Clin Oncol. 2020;17:493-503.
- National Cancer Institute Surveillance, Epidemiology, and End Results Program. SEER Cancer Stat Facts: Leukemia—chronic lymphocytic leukemia. Accessed July 28, 2024. https://seer.cancer.gov/statfacts/html/clyl.html
- Burger JA, Tedeschi A, Barr PM, et al. Ibrutinib as initial therapy for patients with chronic lymphocytic leukemia. N Engl J Med. 2015;373:2425-2437.
- Jain N, Keating M, Thompson P, et al. Ibrutinib and venetoclax for first-line treatment of CLL. N Engl J Med. 2019;380:2095-2103.
About the Author
Jayastu Senapati, MBBS, MD, DM, is an assistant professor in the Department of Leukemia at The University of Texas MD Anderson Cancer Center, Houston.