Clinical trials are research studies that test how well new medical approaches work in people.1 Oncology clinical trials are used to test new methods to detect, prevent, and treat cancer—these different types of trials are known as screening trials, prevention trials, and treatment trials, respectively.2
Clinical trials are essential for advancing our understanding and treatment of cancer. They provide a controlled environment for evaluating the effectiveness of new drugs and therapies relative to established standard-of-care treatments. Through the rigorous testing and evaluation of new treatments, clinical trials ensure that the most promising strategies become available to patients and ultimately become an enhanced standard of care.
Nearly every treatment used today was previously evaluated in a clinical trial. In that sense, yesterday’s trials enabled today’s treatments, and today’s trials will enable the treatments of tomorrow. This is a continuous process of careful scientific research and improvement, ensuring that scientific progress benefits each successive generation of patients.
Through rigorous testing and evaluation, clinical trials ensure that the most promising strategies become available to patients and ultimately become an enhanced standard of care.
Debunking Myths About Clinical Trials
If virtually all treatments used today were once clinical trials, why is there hesitancy and mistrust surrounding clinical trials? Should patients with cancer and their medical teams actively consider clinical trials? And if so, when should they explore their clinical trial options? Myths and misconceptions can deter individuals from participating in potentially life-saving research. Providing accurate information helps to break down barriers, allowing more patients to benefit from the innovative treatments clinical trials offer. The following information may help you decide if participating in a clinical trial is right for you.
Myth #1: General Mistrust of Clinical Trials: “I don’t want to be a guinea pig.”
Fear, suspicion, and mistrust are common barriers that prevent patients, particularly those from underrepresented populations, from participating in clinical trials. These concerns can stem from historical events in which unethical research was conducted before modern-day protections were put in place or from the anxiety trial participants feel about receiving an unproven treatment.
The reality is that clinical trials undergo a rigorous regulatory oversight process that includes several steps before a clinical trial can even begin:
- FDA Review: Any new drug goes through extensive research and testing before researchers submit a request to run a clinical trial via an Investigational New Drug application3 with the FDA. The FDA examines preclinical data, including laboratory and animal studies, before granting permission for human trials to proceed.
- IRB Review: An Institutional Review Board (IRB) then reviews the trial protocol to ensure it protects participants’ rights, safety, and privacy. The IRB is an independent committee made up of medical professionals, ethicists, and community members who evaluate the ethical aspects of the trial and ensure its compliance with ethical standards.
- Informed Consent: Informed consent is a critical process that ensures participants understand the risks, benefits, and responsibilities of participating in a clinical trial. Before joining a trial, potential participants are given detailed information, including:
- The purpose of the trial
- The treatments or interventions involved
- Possible risks and benefits
- How the trial will be conducted
- Participants’ rights, including the right to withdraw from the trial at any time
Participants must voluntarily agree to these terms, and their consent is documented in writing. The informed consent process is ongoing, and participants can ask questions or raise concerns at any time during the trial.
In addition to becoming familiar with all these safeguards, patients should speak with their medical team and ask lots of questions while thoroughly researching treatment options. These are all important steps to increase your trust of the clinical trial process.
Myth #2: Clinical Trials Are Only a Last Resort
Many patients do not consider clinical trials early in their treatment journey. A common myth is that trials should be considered only after every other treatment regimen has failed. In reality, the National Comprehensive Cancer Network (NCCN) Guidelines for Patients recommend considering clinical trial options at each decision point in a patient’s treatment journey: “Everyone with cancer should carefully consider all of the treatment options available for their cancer type, including standard treatments and clinical trials. Talk to your doctor about whether a clinical trial may make sense for you.”4
Because some clinical trials are available only for patients who have not yet had other forms of treatment, waiting until other treatment options have been exhausted can significantly limit your clinical trial options. That’s why the NCCN recommends, “Don’t wait for your doctor to bring up clinical trials. Start the conversation and learn about all of your treatment options.”4
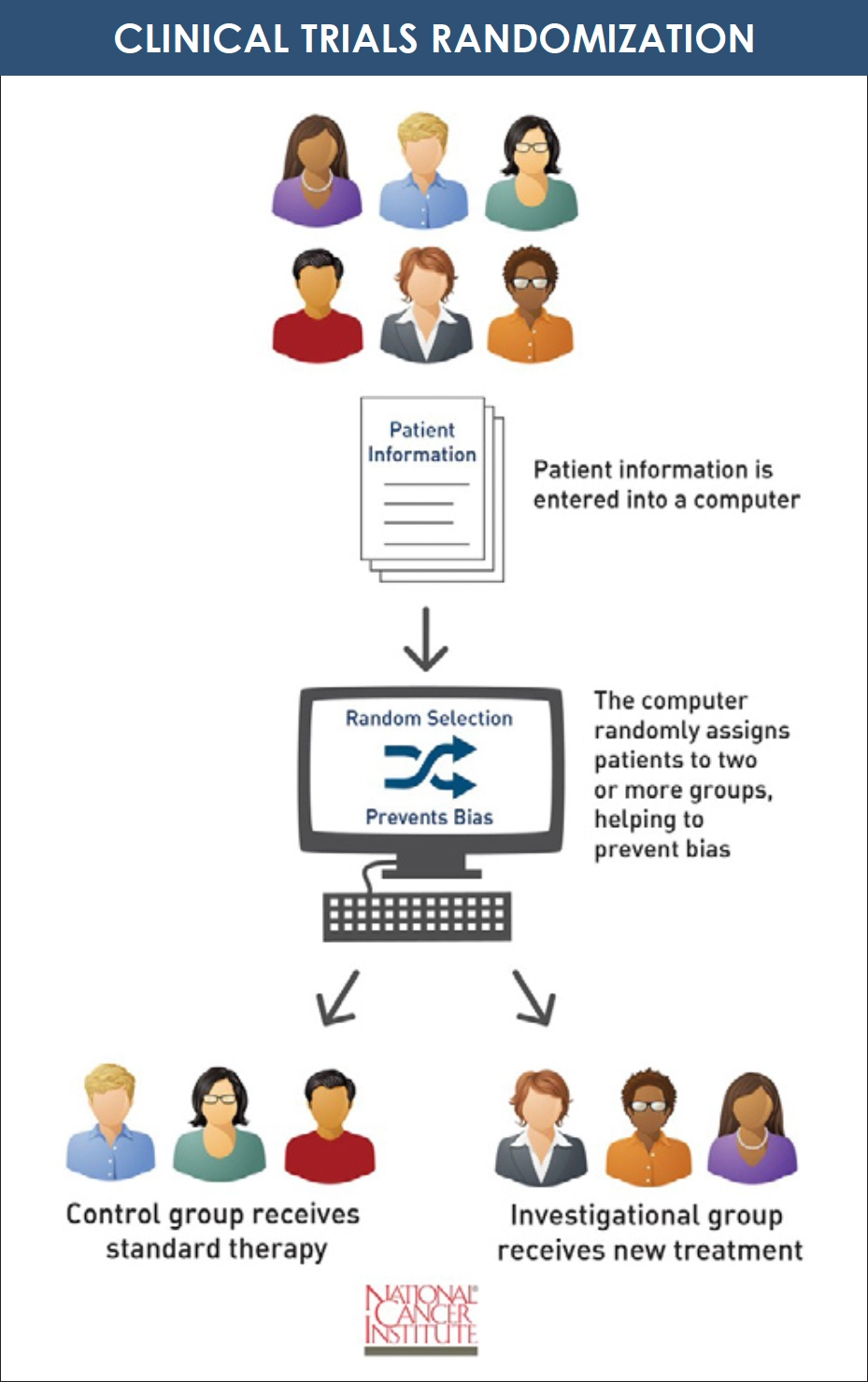
Myth #3: I’ll Only Receive a Placebo, or I’ll Be Randomized Into Getting No Treatment
A commonly expressed concern about clinical trials is that the patient will receive only a placebo—an inactive substance or “sugar pill” with no therapeutic benefit—instead of receiving medicine to treat the cancer. The NCCN clarifies, “Placebos (inactive versions of real medicines) are almost never used alone in cancer clinical trials. It is common to receive either a placebo with a standard treatment or a new drug with a standard treatment. You will be informed, verbally and in writing, if a placebo is part of a clinical trial before you enroll.”4
Randomization is a related process that isn’t well understood. The National Cancer Institute defines randomization as “the process by which participants in clinical trials are assigned by chance to separate groups that are given different treatments or other interventions. Neither the researcher nor the participant chooses which treatment or intervention the participant will receive.”5 Randomization is often part of a blinded study in which the patients and sometimes the doctors don’t know which treatment is being administered. The purpose of randomization and blinded studies is to remove any bias from the study and ensure scientific accuracy of results.
The reality is that all cancer patients will receive at least the treatment that is the standard of care for their particular cancer. They may also receive the treatment being evaluated. Importantly, oncology patients will receive treatment, even in a randomized trial. It may just be the standard-of-care treatment they would have received had they not been enrolled in the trial.
Myth #4: Clinical Trials Are Too Expensive, or There Are Too Many Barriers for Me to Participate
Another common myth surrounding clinical trials is that they are too expensive or come with significant logistical challenges. There are several things every prospective trial participant should know:
What the Trial Sponsor Covers: The trial sponsor, such as a pharmaceutical company or research institution, typically covers the cost of the experimental treatment, diagnostic tests, and certain procedures directly related to the trial. This ensures that participants do not have to pay for the new treatment being studied or additional medical care specifically required by the trial.
What Insurance Covers: Insurance usually covers routine patient care costs during clinical trials, such as doctor visits, hospital stays, and standard tests or procedures unrelated to the experimental treatment. These are typically services the patient would receive as part of their regular care, even if they were not in a trial.
Resources Available to Fill Any Gaps: Financial concerns are often a perceived barrier for people to participate in trials, but many clinical trials provide resources to help address gaps that insurance or the sponsor may not cover, such as travel and lodging assistance, childcare, and even stipends for time spent participating. Patient advocacy groups and trial coordinators often connect patients with these resources to minimize out-of-pocket expenses.
Lack of Transportation/Financial Burden: Some trials offer transportation assistance or reimbursement to ease the burden of traveling to trial sites. This helps remove the financial or logistical barrier for patients who might otherwise be unable to participate because of transportation challenges or costs.
Interference With Work/Family Responsibilities: Clinical trial coordinators are often flexible in scheduling treatments and appointments to accommodate work or family commitments. Some trials may provide compensation for lost wages or offer services like childcare to help balance personal responsibilities with participation in the trial.
Burdensome Procedures: Although some clinical trials involve complex or time-consuming procedures, many sponsors strive to minimize the burden on participants by using remote monitoring or telemedicine or by reducing the frequency of visits to trial sites. In some cases, less invasive alternatives are offered to make participation more manageable.
Strict Eligibility Criteria: Each clinical trial has a set of eligibility criteria that must be met for a patient to participate. These requirements often include factors such as age, general health level, type and stage of cancer, and prior treatments. Criteria for a particular trial can be detailed and extensive, and trial enrollment often restricts participants whose overall health is poor. This is one reason why exploring clinical trial options at every treatment decision point is so important—waiting until a trial is your last resort can mean a trial is no longer an option. Proactively ask your doctor to help identify a trial for you, and/or reach out to advocacy organizations such as the National Cancer Institute (1-800-4-CANCER) or other organizations, to learn what options you have.
Language/Cultural Barriers: Patients from different cultural backgrounds or those who speak limited English may find clinical trials intimidating or inaccessible. To encourage diverse participation, hospitals are increasingly offering culturally sensitive information in multiple languages to explain trials in a way that is clear and understandable to a range of patients.
Myth #5: Misconception That Clinical Trials Aren’t Appropriate for Serious Illnesses
There is a common misconception that clinical trials are only for minor conditions or early-stage illnesses, leading patients with serious diseases like advanced cancer to believe they are not eligible. However, clinical trials are designed for a wide range of illnesses and patients, including those with serious or life-threatening conditions.
For patients with serious illnesses, clinical trials can offer access to cutting-edge treatments that may not be available through standard care. These trials often focus on experimental therapies that could provide new hope for improving outcomes, extending survival, or managing symptoms. Participating in a trial might offer patients a unique opportunity to receive innovative treatments tailored to their specific condition, potentially leading to breakthroughs in their care.
Ultimately, clinical trials play a crucial role in advancing medical knowledge and improving treatment for serious illnesses, making trials highly appropriate for those facing significant health challenges.
It's a Joint Decision
When considering participation in a clinical trial or any treatment plan, it’s important to remember that it’s a joint decision. While doctors provide expertise and guidance, you need to advocate for yourself by asking questions, understanding the options, and ensuring that your voice is heard.
The decision-making process should be a collaborative effort in which you work with your doctor to weigh the risks and benefits. Seeking a second opinion is also a good practice if you’re uncertain or want additional input.
It is not the doctor’s decision to be made in isolation, nor should it be entirely yours—the best outcomes result when both parties arrive at a decision together. Making these decisions can be overwhelming, so including your family and support system will help. Having a village of support is crucial for navigating the complexities of treatment and ensuring that your choices align with your personal values and needs. This is an important decision, and knowing all your options, including clinical trials, will empower you to make an informed decision.
References
- National Cancer Institute. What are clinical trials? Accessed October 28, 2024. www.cancer.gov/research/participate/clinical-trials/what-are-clinical-trials
- National Cancer Institute. NCI’s clinical trials programs and initiatives. Accessed October 28, 2024. www.cancer.gov/research/infrastructure/clinical-trials
- FDA. Investigational New Drug (IND) Application. Accessed October 28, 2024. www.fda.gov/drugs/types-applications/investigational-new-drug-ind-application
- National Comprehensive Cancer Network. Guidelines for patients. Clinical trials. www.nccn.org/patientresources/patient-resources/resources-for-patients-caregivers/clinical-trials
- National Cancer Institute. Dictionary of cancer terms. Accessed October 28, 2024. www.cancer.gov/publications/dictionaries/cancer-terms/def/randomization