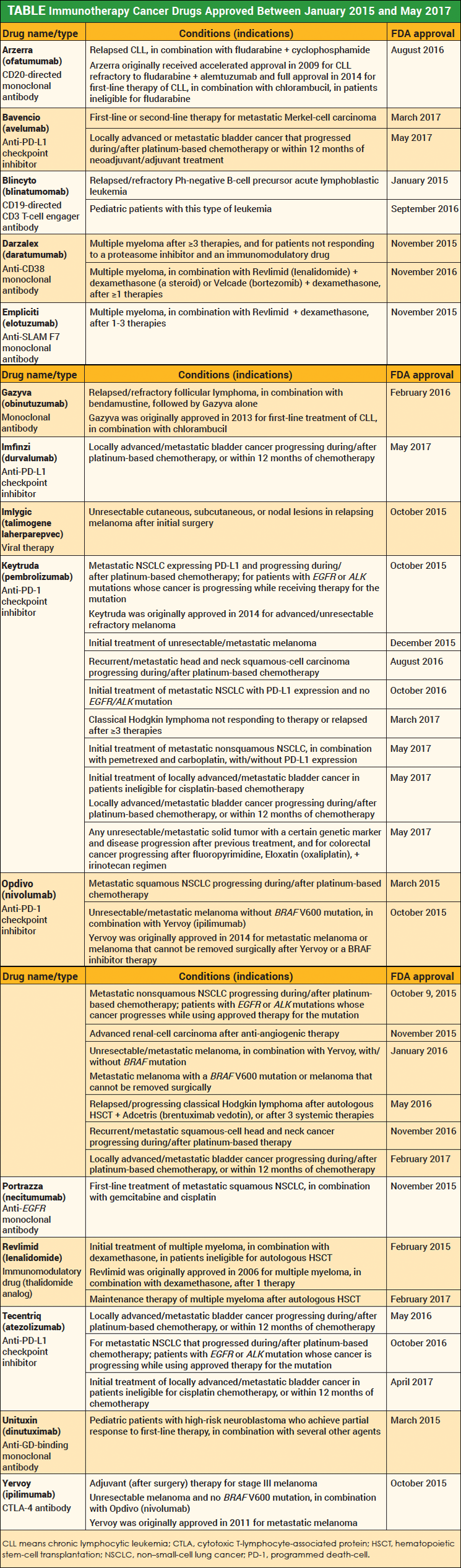
Immunotherapies have been approved by the FDA for more than a decade, but the past 2 years stand out as the period when immunotherapy became a familiar concept in the United States, with the explosion of new therapies that involve the immune system approved by the FDA for many types of cancer.
The Table highlights cancer immunotherapy drugs approved by the FDA between January 2015 and May 2017, focusing on anti-PD-1 and anti-PD-L1 checkpoint inhibitors; antibodies such as monoclonal antibodies and anti-CTLA-4 antibodies; immunomodulatory drugs; and viral cancer therapies.