Perhaps you have been on chemotherapy and are annoyed by its lingering side effects. When can you live normally again? (We agreed to sign on as cancer patients, not martyrs.) Perhaps your treatment regimen included immunotherapy as well. How did that influence the outcome of your disease as well as treatment side effects? What combinations of chemotherapy and immunotherapy now offer the greatest hope for knocking out a cancer while reducing those harsh side effects?
The “Magic Bullets” of Paul Ehrlich
For us to answer those last two questions, it is necessary to look back to the state of medical science’s human disease research a century ago. German researcher Paul Ehrlich (1854-1915) was a farsighted medical pioneer who hypothesized that disease-causing cells could be specifically targeted for their vulnerability to certain chemicals. The burgeoning German synthetic dye industry was teeming with organic chemists who were able to supply Ehrlich with thousands of new chemicals to test as drugs. Ehrlich used the term “magic bullets” to describe substances that would specifically kill pathogens (disease-causing cells) and spare normal cells.1,2 Recall that his research occurred more than a generation prior to antibiotics or any useful anticancer drugs.
Immunological concepts would occupy Ehrlich's latter thinking and would come to be a vital component in today's ADCs, a century later.
Later Ehrlich would coin the term “chemotherapy” to describe the functions of these magic bullets. Toward the end of his life, he imagined a time when special magic bullets would be found to treat cancers. Ehrlich shared the Nobel Prize for Medicine in 1908 for work he had done in 1896 successfully treating common infectious diseases of that era. Ironically, immunological concepts would occupy Ehrlich’s latter thinking and would come to be a vital component in today’s ADCs, a century later.2
ADCs
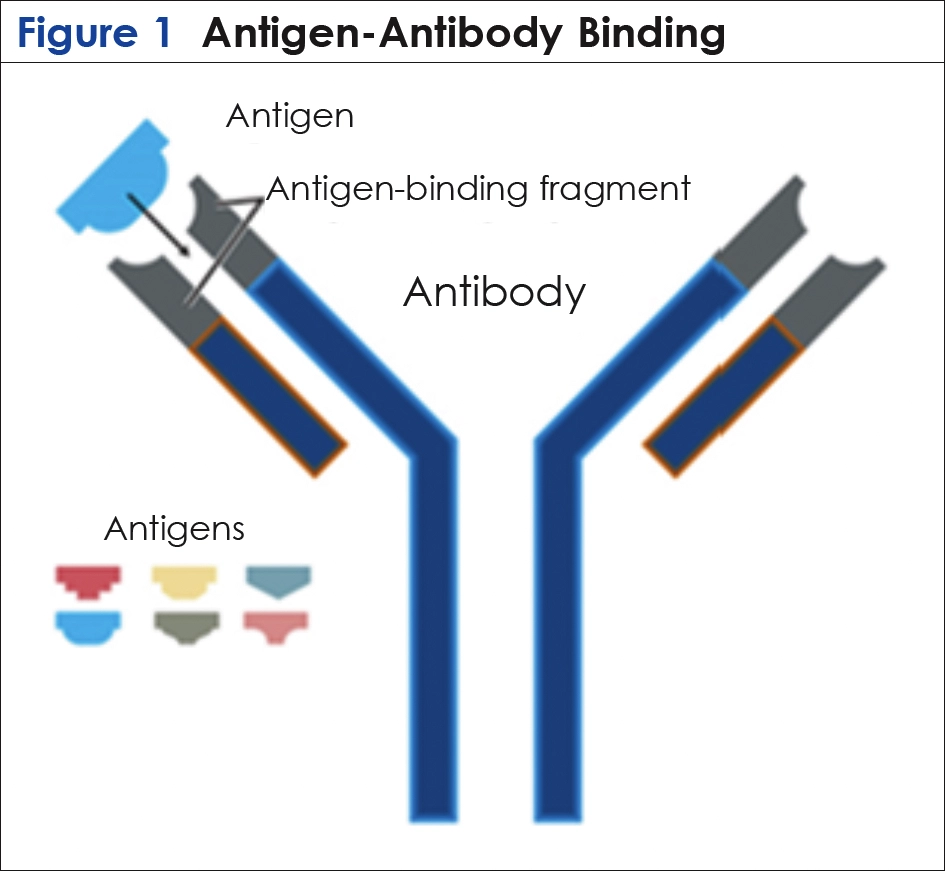
The modern concepts of ADCs, inspired by the “magic bullet” dream of Ehrlich, involved binding a chemotherapeutic drug to an antibody, a means to target the drug toward cancer cells more precisely. Antibodies are natural proteins synthesized by the immune system that bind to specific antigens—molecules on foreign cells that elicit an immune response and bind antibodies stimulated by it with a “lock-and-key” specificity. Antibodies are large Y-shaped proteins that come in numerous highly specific types and are capable of binding to a nearly incalculable number of specifically matched foreign antigens. (It is this antigen-antibody interaction that is the basis for vaccine development.)
This antigen-antibody binding occurs at the top of the Y (Figure 1), triggering further activity by the immune system to destroy the cancer cell bearing the antigen and clearing it from the body. Researchers in the late 1950s proposed chemically linking cancer antigen-specific antibodies with anticancer drugs, thus theoretically providing targeted “smart chemotherapy.” This new construct would in theory avoid collateral damage to nearby normal cells, the cause of those dreaded side effects associated with standard chemotherapy.3-5
A Shaky But Familiar Start
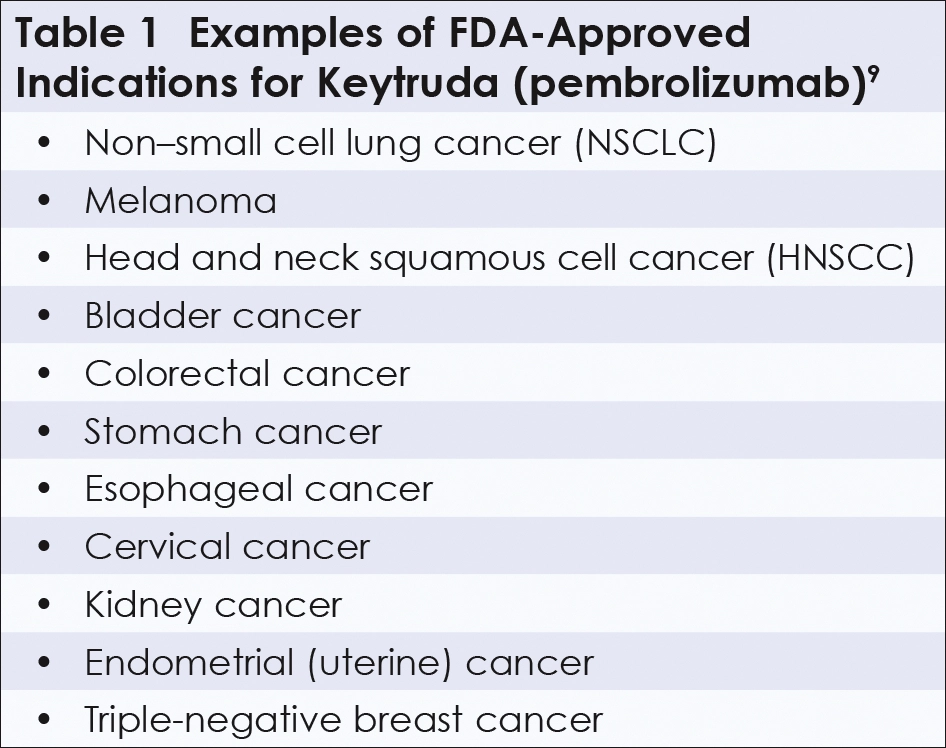
Like the history of cancer chemotherapy or radiotherapy during the 20th century, the history of ADC treatment of human cancers is full of fits and starts. The first clinical trials during the late 1950s through the 1980s were disappointing and still manifested those nasty side effects. But novel biotechnology developed in 1975 revived the ADC concept. At Cambridge University, Georges Köhler and César Milstein fused an antibody-secreting cell with a cancer cell, resulting in a hybridoma, a new cell type that could secrete previously unattained amounts of highly specific antibodies.1,6 (Köhler and Milstein, along with Niels Jerne, were awarded the 1984 Nobel Prize in Medicine.) In spite of their cancerous parentage, these hybridoma-produced antibodies—known as monoclonal antibodies—were not malignant and proved a boon to all immunology. A widely applied therapeutic monoclonal is Keytruda, the trade name for the pembrolizumab monoclonal antibody, which is now an immunotherapy workhorse familiar to many from commercial TV. The Keytruda antibody has been clinically used to boost a patient’s immune response when used in conjunction with chemotherapy against a wide variety of recurrent or metastatic tumors (Table 1).7-9
The medical world now was anxious to synthesize a clinically useful ADC that included monoclonal antibodies. This ADC has 3 essential components:
- Monoclonal antibody (mAb), a protein shaped like a Y
- Linker, a molecule connecting the stem of a mAb to payload drug molecules
- Chemotherapeutic drug attached to the upright stem of the Y
mAbS Are Known For Tongue-Twister Names
The generic name for a monoclonal antibody is usually a tongue-twister, such as gemtuzumab, trastuzumab, or pembrolizumab. They are easy to spot (but not pronounce) because of the suffix -mab, short for monoclonal antibody. Regarding Enhertu (trastuzumab deruxtecan), for example, trastuzumab-deruxtecan refers to the generic name for the monoclonal antibody, trastuzumab, followed by deruxtecan, the name of the drug (payload) within the ADC, and Enhertu is the trade name. Manufacturers assign their own trade names to these drugs and the antibodies that carry them.
Clinical Victory For ADCs Over Breast Cancer
Herceptin, used to treat breast cancer and first approved by the FDA in 2019, incorporates the anti–breast cancer monoclonal antibody trastuzumab into an ADC.
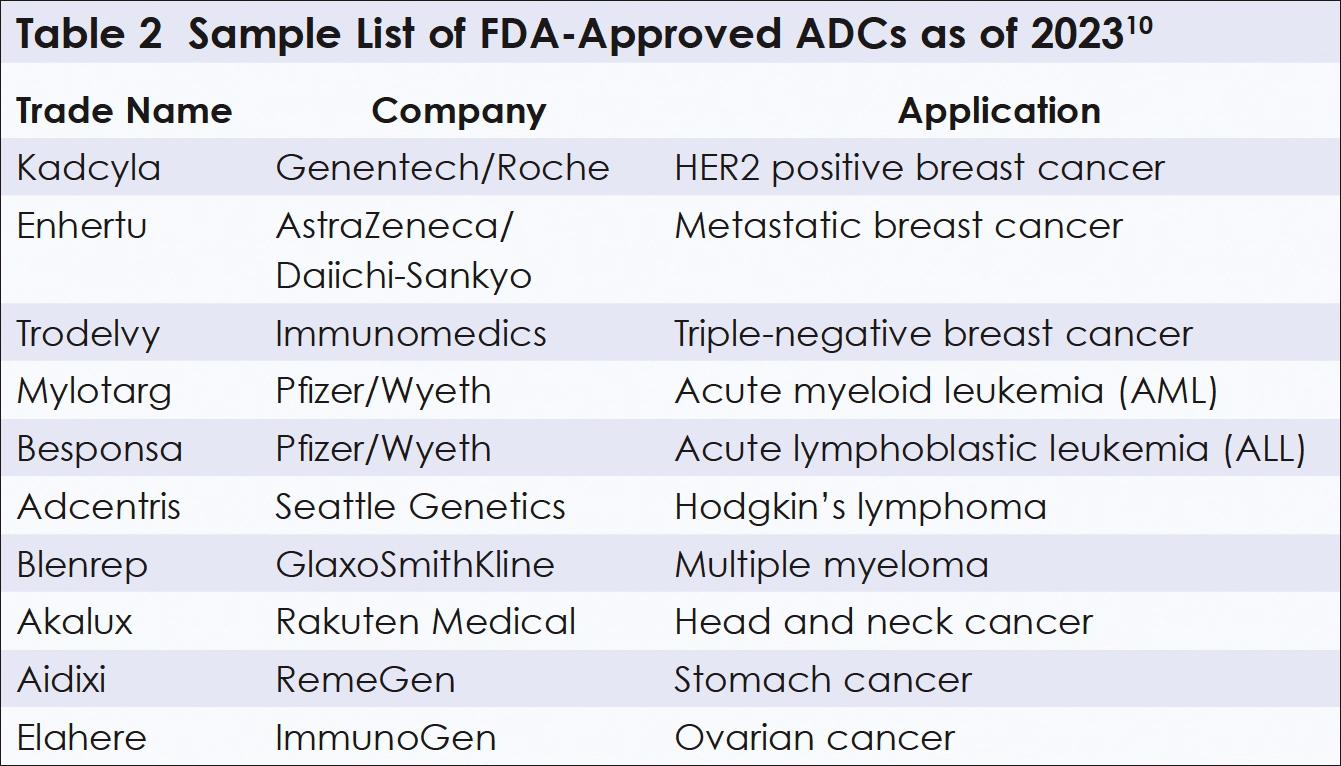
In 2000, the FDA’s first approval for ADC therapy involved gemtuzumab ozogamicin (trade name Mylotarg) for treatment of acute myeloid leukemia.3 Unfortunately, approval was later withdrawn due to severe side effects. Clearly, tweaking had to be done to the selected antibody, the payload, and/or the linker to balance the ADC’s clinical effectiveness and toxicity to normal cells. During the new century, 15 different ADCs have been approved by the FDA since that rocky early clinical trial.10 One of the most successful ones, Herceptin, used to treat breast cancer and first approved by the FDA in 2019, incorporates the anti–breast cancer monoclonal antibody trastuzumab into an ADC.11 Herceptin itself was first developed by biotech giant Genentech (South San Francisco, CA) primarily due to the guidance and persistent lobbying of UCLA’s premier cancer researcher Dennis Slamon.1,12 Herceptin represents a building block in constructing ADCs to treat various cancer types.
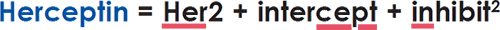
- Why is the monoclonal antibody Herceptin (trastuzumab) effective against breast cancer? When Herceptin homes onto the surface of breast cancer cells, it stimulates several processes that lead to the demise of that cell. These include stimulating a slow process of cellular self-destruction known as apoptosis. It also signals the body’s cellular immune system to release natural killer cells that directly attack the cancer cell.4
- What is HER2? HER2 is a protein that functions as the antigen that specifically binds the monoclonal antibody Herceptin (trastuzumab). The HER2 protein is coded for by the HER2 gene located within the cell’s DNA.1 A gene will code for the production of a protein. Known as a proto-oncogene (pre-cancer gene), the HER2 gene codes for a normal growth factor receptor (HER2 protein) that is associated with a cancer only when it is present in large amounts. This happens when there are multiple copies of that HER2 gene or an overactive gene from mutations. This will produce far too much HER2 protein and will then become designated an oncogene (cancer gene).
- What is the advantage of using an ADC to deliver the payload drug? The forked end of the Y-shaped antibody targets the cancer cell by binding to a specific breast cancer antigen, HER2. Oncologists already recognized what Herceptin alone could do. The key breakthrough in the Herceptin ADC story is the delivery of Herceptin’s payload chemotherapeutic drug (attached to the upright stem of the Y) to within the target cell, thus avoiding any collateral damage to nearby cells. This double whammy from the ADC was truly “smart chemotherapy”: the monoclonal antibody binds specifically and tightly to the targeted HER2 cancer antigen—stimulating the immune system. This is followed by delivery of the payload drug inside the cancer cell, thus avoiding collateral damage to normal cells, the cause of those dreaded side effects.
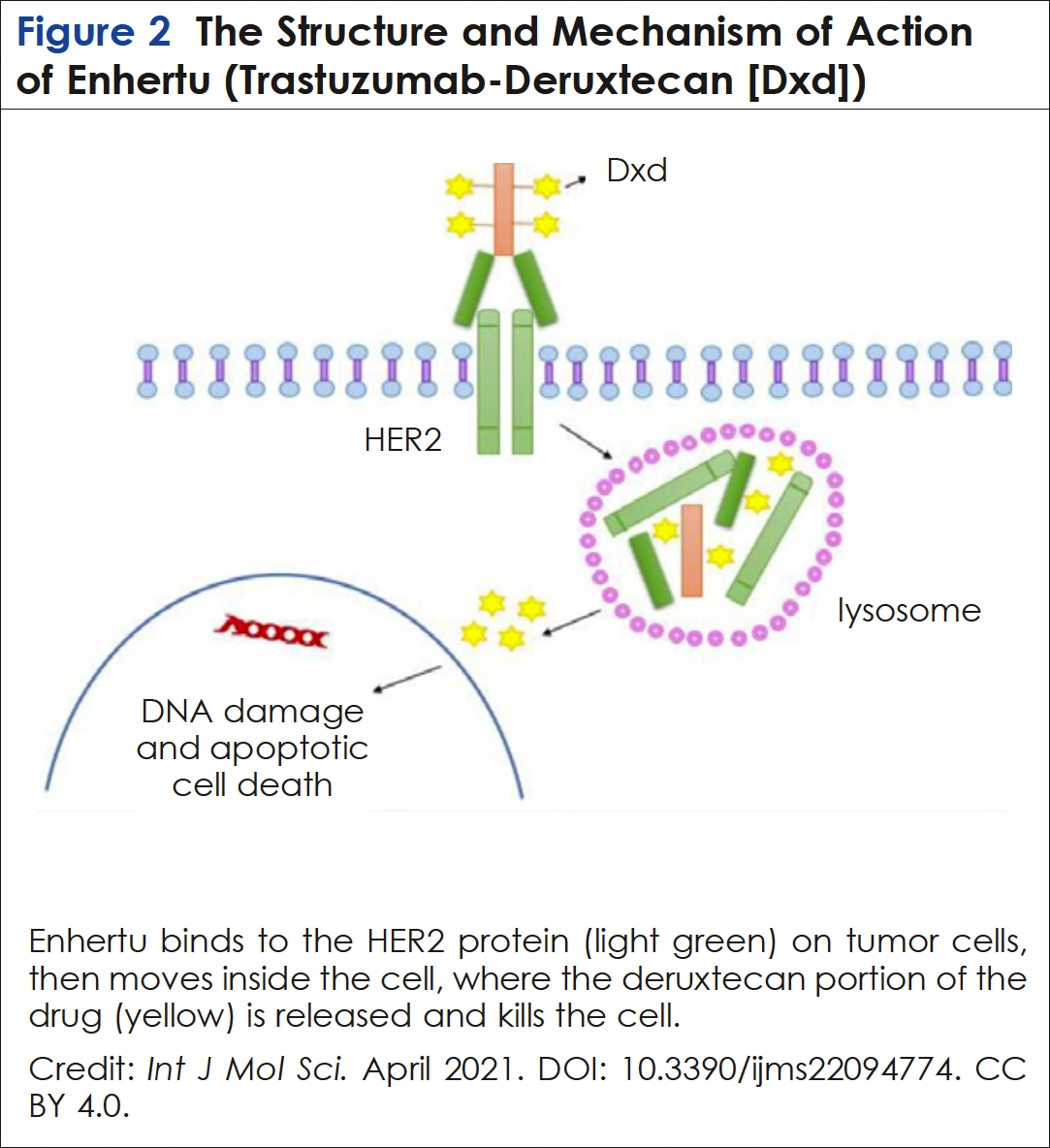
- Why was the Herceptin ADC more successful compared with earlier attempts? Synthetic chemists came to the rescue. Earlier attempts at ADC therapy were problematic because some ADC conjugates were not taken up properly into the cancer cell or, if they had been, were not properly broken down to release the payload drug. That resulted in disappointing clinical effectiveness and/or more toxic side effects. The ideal ADC had to be taken into the cancer cell by engulfing it within a membrane. This structure then would fuse with a membranous sack of digestive enzymes inside the cancer cell known as a lysosome. The creators (AstraZeneca- Daiichi Sankyo) of this new ADC, trastuzumab deruxtecan under the trade name Enhertu, devised a linker that would be neatly cleaved by the lysosome’s enzymes (as would the antibody carrying it), freeing the payload drug to attack the target5,6 (Figure 2—in this case the breast cancer cell’s DNA—stabilizing enzymes known as topoisomerases.13
Good Clinical Results Across the Spectrum of Breast Cancers
HER2-positive breast cancers, the most aggressive ones, constitute about 20% of all cases. Surprisingly, Enhertu was also an effective ADC against the majority of cases that are apparently HER2-negative! HER2 detection methods have grown more sensitive in recent years. Those HER2-negative cases may actually carry a minimal amount of HER2, enough for the high-affinity monoclonal antibody on Enhertu to bind to it, be absorbed into the cell, and deliver its payload within the cancerous cell.5,13 Enhertu has also been clinically effective against HER2-positive cancers of the lung, stomach, and colorectum.14 Other promising ADCs have been clinically applied to various tumors (Table 2).3,10
ADCs in Future Cancer Therapies
Radioisotopes could also be linked to a monoclonal antibody to deliver “smart radiotherapy,”… minimizing radiation damage to nearby normal cells.
Dozens of new ADCs with variations in their payloads, in the role of a “pharmaceutic Trojan horse,”14 are currently undergoing clinical trials.14,15 More exotic variations in this payload will entail compounds that stimulate the human immune system. Radioisotopes could also be linked to a monoclonal antibody to deliver “smart radiotherapy,” as radio-conjugates, more selectively to the patient’s tumor, again minimizing radiation damage to nearby normal cells. Given clinical breakthroughs and budget increases earmarked by biotech and pharmaceutical companies for ADC research, the number of ADCs synthesized for human clinical trials will likely grow geometrically.3,5 A great mix-and-match of monoclonal antibodies and drug payloads may enable oncologists to reduce chemotherapy dosage without sacrificing drug efficacy. Drugs too toxic for stand-alone chemotherapy could be delivered using the targeted ADC approach. Precision medicine, in which therapies are tweaked toward individuals, not generalized categories of patients, will dominate future cancer treatments. New genetic technologies are identifying new cancer-specific antigens against which monoclonal antibodies can be produced and readily linked to anticancer drugs, forming 21st century versions of Ehrlich’s magic bullets. Stand-alone chemotherapy may become a memory carried by older generations of patients. Personally, I would prefer to change the designation ADC to mean “Antibody-Directed Chemotherapy.” In any case, may such smart chemotherapy14 help to fulfill the oncologist’s mantra of more cures, less toxicity, enhancing both the quantity and quality of life for cancer patients.
References
- Mukherjee S. The Emperor of All Maladies: A Biography of Cancer. Simon & Schuster; 2010.
- The Nobel Prize in physiology or medicine 1908. Accessed June 13, 2024. www.nobelprize.org/prizes/medicine/1908/ehrlich/biographical/
- Fu Z, Li S, Han S, et al. Antibody drug conjugate: the “biological missile” for targeted cancer therapy. Signal Transduct Target Ther. 2022;7:93.
- Valabrega G, Montemurro F, Aglietta M. Trastuzumab: mechanism of action, resistance and future perspectives in HER2-overexpressing breast cancer. Ann Oncol. 2007;18:977-984.
- Madhusoodanan J. Targeting cancer, sparing patients. Scientific American. 2024;330:36-41.
- Köhler G, Milstein C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature. 1975;256:495-497.
- Doyle C. First-line treatment with Keytruda doubles the time without disease progression in patients with MSI-H or dMMR metastatic colorectal cancer. Conquer. Accessed June 13, 2024. https://conquer-magazine.com/issues/special-issues/august-2020-asco-highlights/1339:first-line-treatment-with-keytruda-doubles-the-time-without-disease-progression-in-patients-with-msi-h-or-dmmr-metastatic-colorectal-cancer
- Kwok G, Yau T, Chie J, et al. Pembrolizumab (Keytruda). Hum Vaccin Immunother. 2016;12:2777-2789.
- Keytruda. Prescribing information. Merck. Accessed June 11, 2024. www.keytrudahcp.com/prescribing-information/
- Wang Z, Li H, Gou L, et al. Antibody-drug conjugates: recent advances in payloads. Acta Pharm Sin B. 2023;13:4025-4059.
- Emens L. Breast cancer immunotherapy: facts and hopes. Clin Cancer Res. 2018;24:511-520.
- Slamon D, Eiermann W, Robert N, et al. Adjuvant trastuzumab in HER2-positive breast cancer. N Engl J Med. 2011;365:1273-1283.
- Shastry M, Gupta A, Chandarlapaty S, et al. Rise of drug-antibody conjugates: the present and future. Am Soc Clin Oncol Educ Book. 2023;43:e390094.
- Tarantino P, Pestana R, Corti C, et al. Antibody-drug conjugates: smart chemotherapy delivery across tumor histologies. CA: Cancer J Clin. 2021;72:165-182.
- NJ Bio. Recent advances in ADCs. Updated May 6, 2024. Accessed June 13, 2024. https://njbio.com/antibody-drug-conjugates/
About the Author
Garry M. Marley, PhD, currently teaches within the faculty of the Department of Microbiology
& Molecular Genetics at Oklahoma State University. He holds a PhD in molecular biology and
microbiology from the University of Maryland at Baltimore and an MSc in pathobiology from
the Johns Hopkins Bloomberg School of Public Health. Last year he was successfully treated for a head and neck cancer with chemoradiotherapy at the Stillwater Cancer Center in Oklahoma.
He can be reached at